LSU Chemist Investigates Oxidized Cholesterol Molecule as Potential Therapy for Colorectal Cancer
March 11, 2026

LSU chemistry professor Dr. Fatima Rivas studies bioactive molecules with potential for future cancer therapies.
Colorectal cancer is one of the most common and deadly cancers in the United States. In Louisiana, the burden is especially severe: the state consistently ranks among the highest in the nation for both incidence and mortality, with many patients diagnosed at later stages, when treatment options are more limited.
Rates are also rising among younger adults, making the search for new treatments increasingly urgent. While screening and prevention remain essential, scientists are also investigating the molecular processes inside our cells that influence how cancers grow — and how the body’s own signaling molecules might be harnessed to fight disease.
Among the many chemical reactions occurring in the human body are those that synthesize cholesterol. Along this pathway, intermediate molecules are formed and can sometimes become oxidized during cellular stress. One such oxidized intermediate has recently captured the attention of researchers at LSU.
“One of the challenges in chemistry and biology is that we still don’t fully understand many of the molecules that exist in our own bodies,” said LSU chemist Dr. Fatima Rivas. “Some were identified decades ago, but their function is still unclear.”
Rivas and her research team are now studying whether one of these understudied molecules could help fight colorectal cancer. The project recently received pilot funding through the Seed Grants program of the Louisiana Cancer Research Center, which supports early-stage research aimed at developing new cancer therapies.
A Discovery Inspired by Natural Compounds
Rivas’ lab studies natural products — molecules produced by plants, fungi, and other organisms that can influence biological processes. Many modern medicines, from antibiotics to cancer therapies, were originally inspired by compounds discovered in nature.
While studying bioactive molecules produced by mushrooms, the LSU team became interested in a class of compounds known as endoperoxides. These molecules contain a highly reactive oxygen–oxygen bond that can break open inside cells, releasing chemical species capable of disrupting essential cellular functions.
As researchers examined these mushroom-derived compounds, they noticed that a molecule with a similar structure could also arise from compounds already present in the human cholesterol pathway.
That observation led them to an oxidized cholesterol intermediate — a molecule that resembles cholesterol but contains a peroxide group. On paper, its chemical structure resembles that of cholesterol, but its three-dimensional shape is bent in ways cholesterol is not.
Unlike dietary oxidized cholesterol, this compound forms inside the body as a short-lived intermediate in cholesterol biosynthesis and is typically present at very low levels in the bloodstream. Because it retains much of cholesterol’s structure, cells may recognize and take up the molecule in similar ways.
Dr. Rivas describes the process as a kind of molecular “Trojan horse.”
“The cells recognize the molecule as something similar to cholesterol and bring it inside,” she explained. “Once inside, that peroxide group can become activated and disrupt processes that the cell depends on.”
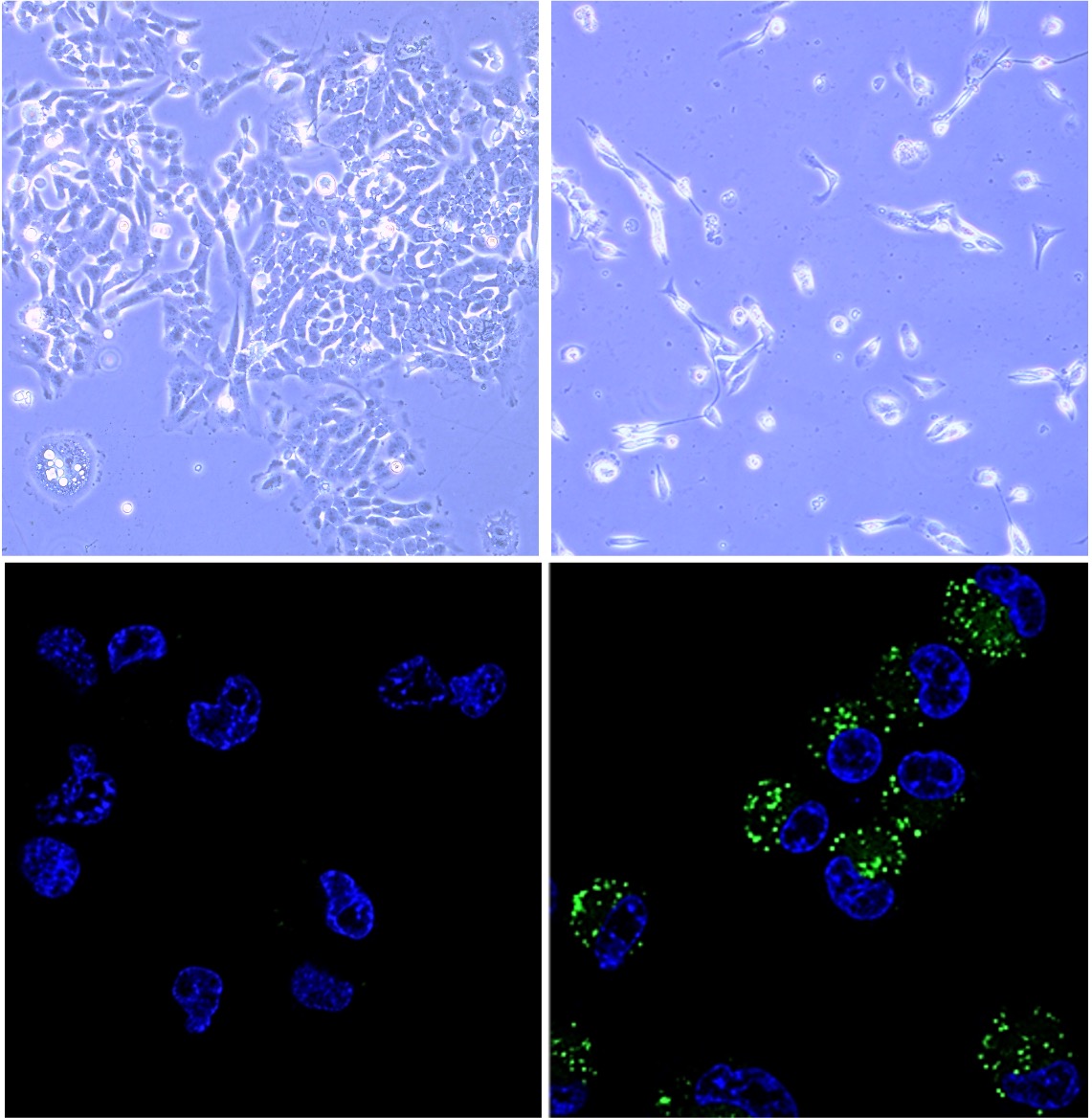
Microscope images reveal how the experimental molecule affects colorectal cancer cells. Untreated cells (top left) remain healthy, while treated cells show structural changes consistent with cell death (top right). A fluorescent version of the molecule (bottom right) allows researchers to see how the compound is taken up by cells, compared with untreated controls (bottom left).
To study the molecule in detail, the LSU team synthesized it in gram-scale quantities, allowing them to test its biological activity in laboratory models. The researchers evaluated the compound across several colorectal cancer cell lines, including HCT116, HT-29, Caco-2, and RKO.
The results were promising.
At relatively low concentrations, the compound slowed cancer cell growth and triggered apoptosis, a controlled form of programmed cell death. Importantly, the molecule showed selectivity: it was more harmful to colorectal cancer cells than to normal cells, including liver cells and immune cells.
That selectivity may reflect fundamental biological differences between healthy and cancerous tissue.
Cancer cells grow and divide rapidly, placing heavy demands on systems that repair damage and remove faulty proteins. Early evidence suggests the oxidized cholesterol compound interferes with these protein quality-control pathways — particularly those that tag damaged proteins for removal through a process known as ubiquitination.
“Cancer cells depend heavily on certain pathways because they are dividing so quickly,” Rivas said. “If you disrupt those pathways, the cells cannot keep up with the stress and eventually die.”
The compound also does not appear to cause widespread oxidative damage. Instead, researchers observed targeted cellular stress and disruptions to membrane function — signs that the molecule may interfere with specific regulatory processes inside cancer cells.
New Possibilities for Future Cancer Therapies
If further studies confirm its promise, the oxidized cholesterol compound could represent a new strategy for targeting colorectal cancer by exploiting the metabolic differences between healthy and cancerous cells.
For Rivas, the project also illustrates how discoveries in natural products chemistry can reveal unexpected insights into human biology.
“Nature has evolved molecules that interact with biological systems in very specific ways,” she said. “Sometimes those molecules inspire us to look again at compounds that already exist in our own metabolism.”
The research is still in its early stages, with experiments currently limited to cancer cells grown in the laboratory. The team is now developing chemical derivatives designed to improve the compound’s stability and selectivity, while running biochemical “pull-down” experiments to identify the precise proteins it interacts with inside the cell.
Understanding those targets will be critical for determining how the molecule works — and whether it could eventually be developed into a therapeutic candidate.
If the results continue to hold, the researchers hope to move toward pre-clinical studies and eventually pursue larger federal funding.
“This is a difficult cancer subtype,” Rivas said. “But with creativity and careful research, we can begin to uncover new possibilities.”