Tiny Worms, Big Clues: Mapping How Gut Microbes Shape Metabolism
February 23, 2026
Metabolic disorders such as type 2 diabetes are rising worldwide, contributing to kidney, liver, and cardiovascular disease. While diet and genetics are well-known risk factors, scientists are increasingly recognizing another powerful force shaping metabolic health: the trillions of microbes living in the gut.

Assistant Professor Fan Zhang investigates how gut microbes shape metabolism and insulin signaling in the LSU Department of Biological Sciences.
“Bacteria are not just pathogens,” said LSU biological scientist Fan Zhang, who recently received his first NIH award for early-stage investigators (R35). “Many actively support host fitness. They break down dietary fiber, produce vitamins and signaling molecules that are essential for physical and mental health.”
In type 2 diabetes, beneficial microbes decline while bacteria associated with inflammation become more dominant. The gut community also shifts toward increased carbohydrate processing, which may further affect metabolic regulation.
But recognizing that imbalance is only the first step. The gut contains hundreds of interacting microbial species whose numbers fluctuate constantly, creating an enormous range of possible community combinations. Scientists are still trying to determine which microbial profiles best support metabolic health — and how signals produced by these microbes travel from the intestine to the brain to influence insulin regulation and whole-body function.
Now Zhang is working to decode this system at the molecular level. His five-year award supports efforts to determine how specific microbial communities shape endocrine signaling — particularly insulin regulation — along the gut–brain axis. To make that possible, his lab turns to a model organism that simplifies the biology without sacrificing relevance.
A Simplified System for a Complex Problem
At the center of this approach is C. elegans, a transparent, microscopic worm with only about 1,000 cells — including 20 intestinal cells and 302 neurons — yet its metabolic signaling pathways are highly conserved with those of humans.
Because the worms can be rendered germ-free at the start of each experiment, researchers introduce precisely defined microbial communities and observe physiological consequences under tightly controlled conditions.
“In humans, hundreds of bacterial species change at once. It’s very hard to know cause and effect,” Zhang said. “With worms, we start from a clean background and build the microbiome step by step.”
Using high-throughput 96-well formats and fluorescent insulin reporters, the lab measures how defined microbial communities affect reproductive output, lifespan, and cellular activity. The worms’ transparency allows researchers to directly visualize insulin signaling in specific gut and neuronal cells — linking microbial composition to endocrine response. Beyond these imaging experiments, the team is building an open-access database cataloging endocrine gene expression in response to individual strains and microbial communities, creating a resource to uncover broader bacterial functions that regulate host endocrine signaling.
Closing the Loop: From Microbiome Selection to
Molecular Signals
The new project builds on Zhang’s earlier discovery that insulin signaling does more than respond to microbes — it helps determine which bacteria colonize the gut. His previous work at Baylor College of Medicine showed that natural genetic variation among wild strains of C. elegans drives the assembly of distinct microbiome types. Variations in insulin signaling pathways influenced enrichment of specific commensal bacteria, including members of the genus Ochrobactrum. Worms that preferentially enriched these bacteria exhibited faster growth rates and larger body size, linking microbiome composition to host fitness.
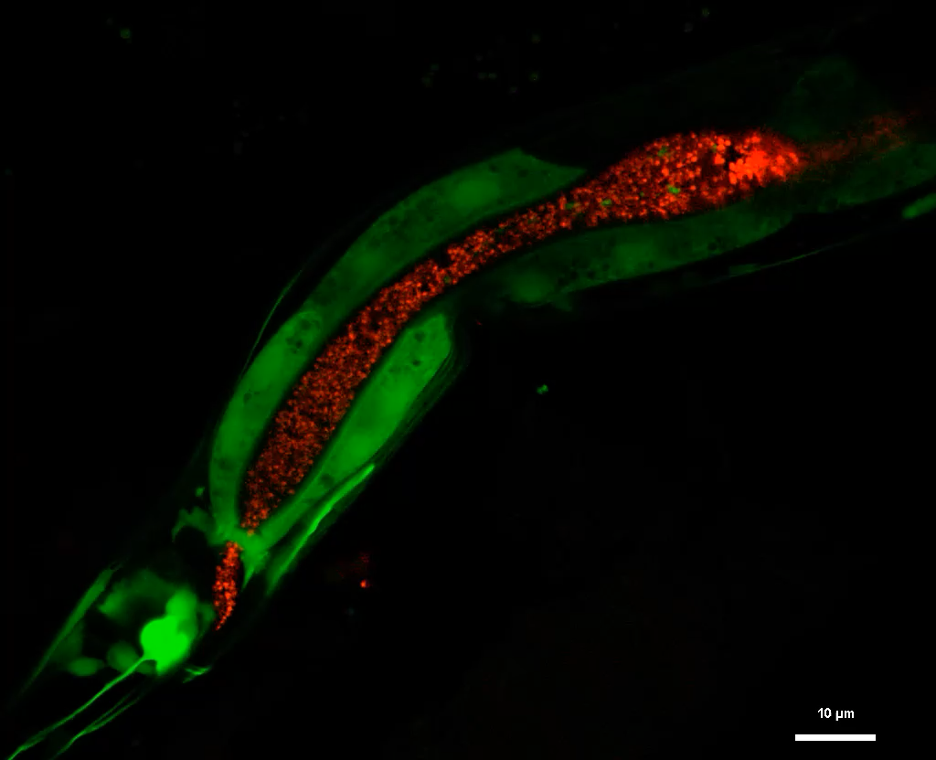
Fluorescent imaging shows insulin expression (green) in neurons and intestinal cells of living worms, alongside gut microbes (red) within the intestinal lumen.
In insulin-resistant worms carrying mutations in insulin receptor genes, that selective enrichment diminished.
“When insulin signaling is disrupted, the worms are not as selective,” Zhang said. “They can’t enrich beneficial bacteria as effectively.”
Together, these findings revealed a bidirectional relationship: host endocrine signaling shapes microbiome composition, and microbiome composition, in turn, correlates with growth and physiological performance. The new project now closes that loop by asking how specific microbes — and the molecules they produce — feed back to regulate insulin signaling and whole-organism physiology.
Early observations highlight how dynamic that relationship can be.
“Even if we start with just one percent of one specific bacterium in the mixture, it can expand to fifty or seventy percent inside the gut,” Zhang said. “That enrichment was surprising.”
The lab now seeks to understand what enables that competitive advantage and which bacterial metabolites activate insulin signaling in intestinal and neuronal cells. Rather than focusing solely on probiotics, Zhang aims to identify postbiotics — defined microbial molecules that directly modulate endocrine pathways. To achieve this, the lab is conducting deeper molecular analyses, including proteomics, metabolomics, and gene expression profiling, to identify specific bacterial metabolites driving the response.
Although certain microbes can briefly dominate under controlled conditions, the microbiome is constantly shifting — making lasting change difficult, especially in humans.
“It’s very hard to permanently change microbiome composition,” he said. “Even when we introduce live bacteria orally, they often disappear after a few days. Targeting the active molecules they produce may provide a more reliable strategy for improving metabolic health in humans.”
Prestigious Pathway Award Recognition
This award is designated a Prestigious Pathway Award at LSU. The distinction includes a $5,000 stipend and recognition at the university’s Gold Standard of Excellence event, which honors faculty achievements that advance LSU’s research mission.