XAS Applications in Environmental Sciences

In the popular everything-automobile-related talk show on public radio a caller enquired about the best method for removal of the light-colored fluffy deposit (fuzz) often found around dead or weak lead-acid automotive batteries. It appears that either of the soft drinks Coke or Dr. Pepper (both contain phosphorus acid) can remove this deposit easily but user preference is cultural, depending on his or her residency above or below the Mason-Dixon line. A detailed search of the scientific literature or the Internet did not yield very specific information about this deposit. Very often it is suggested that the product is high in lead.The efflorescence sometimes observed on lead-acid automotive batteries was studied by X-ray absorption spectroscopy (XAS) and other techniques. The minerals identified are chalcanthite (CuSO4.5H2O) and szomolnokite (FeSO4.5H2O), with a minor amount of poitevinite (Cu, Fe, Zn)SO4.5H2O. Trace amounts of nickel, manganese and chromium, calcium and antimony are detected in the efflorescence. There is very little lead, however.
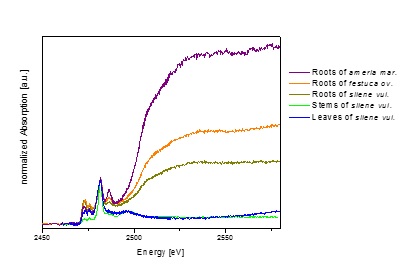
Phytoremediation: How is Pb stored in plants? – Can phytoremediation used with Pb? – the short answer is No.
 |
 |
|
| Many plants cannot tolerate a high concentration of Pb in the ground. For this research plants from a meadow with high Pb concentrations were investigated. One of these plants is sea pink. | When Pb is stored in plants one question is: Is it taken up into the leaves? For most plants we find that is not the case |