Bhattacharya Publishes Paper on Electrocatalysts Generated From Copolymer Templates
May 27, 2021
 BATON ROUGE, LA – LSU Chemical Engineering PhD candidate Deepra Bhattacharya recently
published his first paper as lead author in the publication Small, an academic journal
dedicated to science at the nano- and microscale. He is advised by Assistant Professor
of Chemical Engineering Chris Arges.
BATON ROUGE, LA – LSU Chemical Engineering PhD candidate Deepra Bhattacharya recently
published his first paper as lead author in the publication Small, an academic journal
dedicated to science at the nano- and microscale. He is advised by Assistant Professor
of Chemical Engineering Chris Arges.
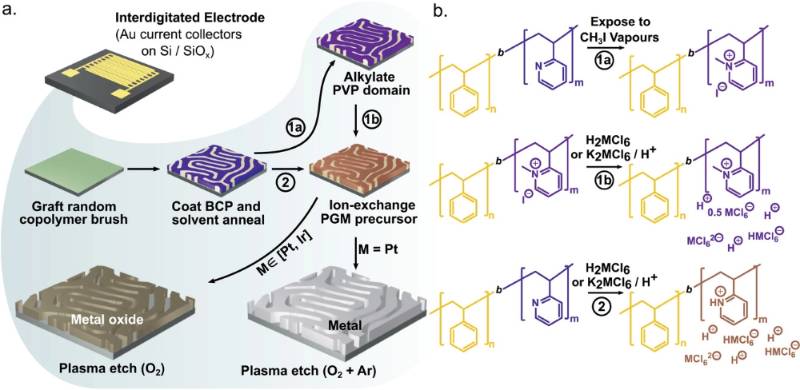
The paper, titled “Electrolysis on a Chip With Tunable Thin Film Nanostructured PGM Electrocatalysts Generated From Self-Assembled Block Copolymer Templates,” centers on Bhattacharya’s research into fabricating electrocatalysts of four distinct geometries using various block copolymers as templates—three being lamellar morphology of different feature sizes and one cylindrical.
The lamellar templates produce structures that look like nanowires, and the cylindrical morphology appears as circular disks. The templated platinum and irdium oxide electrocatalysts—characterized by the Advanced Photon Source synchrotron facility at Argonne National Laboratory, in addition to LSU’s Shared Instrumentation Facility, Nanofabrication Facility, and the Center for Advanced Microstructures and Devices (CAMD)—were found to have near-flawless nanostructure over several square-millimeters of area. The performance of these electrocatalysts was then assessed by examining their propensity to oxidize and evolve hydrogen gas in a lab-on-a-chip setup analogous to an electrochemical hydrogen pump, which is a commercial device used for hydrogen separations and compression.
What Bhattacharya and other Arges Lab group members found were several exciting research avenues and a more efficient experimental approach.
“Electrochemical energy conversion devices, such as fuel cells and electrolyzers, are vital technologies to transitioning to the much-coveted hydrogen economy, where fossil fuels are no longer our primary source of energy,” Bhattacharya said. “This work can help achieve ambitious targets by the U.S. government to reduce greenhouse gas emissions by 50 percent in 2030 [over 2005 levels]. An important research priority for the fuel cell and water electrolysis community is to reduce platinum group metal (PGM) loadings within the devices through better catalyst utilization. PGMs are rare and expensive. Better electrocatalyst utilization can be achieved through nano-scale engineering of the structures and tuning electrocatalyst-electrolyte interfaces.
“We report a simple, inexpensive lab-on-a-chip experimental setup that allows for high throughput experimental analyses that relates electrocatalyst composition and nanostructure, as well as electrolyte and electrode type, to catalytic reactivity. The lab-on-a-chip platform uses 100 times less PGM than traditional experimental setups for gas-phase fuel cell and electrolysis assessments. Conventional methods of testing such materials deploy fundamentally different experimental setups that often lead to findings that do not hold true in real-world applications or involve expensive and time-consuming tests. Our interdigitated electrode platform, which is simple, compact, and inexpensive, is similar in architecture to real-world devices and allows for quick comparative electrochemical analyses of novel materials. This platform hast the potential to substantially speed up materials research in the area and generate data for machine learning activities.”
Click here to read the full paper.
Co-authors on the paper with Bhattacharya were Subarna Kole, chemical engineering PhD candidate at LSU; Orhan Kizilkaya, in the Center for Advanced Microstructures and Devices at LSU; Joseph Strzalka, in the X-ray Science Division at Argonne National Laboratory; Polyxeni Angelopoulou and Georgios Sakellariou, with the Department of Chemistry at the National and Kapodistrian University of Athens in Greece; Dongmei Cao, in LSU’s Shared Instrument Facility; and Arges.
The work was funded by the U.S. Department of Energy Office of Science, Separation Science Program.
Like us on Facebook (@lsuengineering) or follow us on Twitter and Instagram (@lsuengineering).
###
Contact: Joshua Duplechain
Director of Communications